Zoonotic Risk Calculator
Zoonotic Disease Risk Assessment
Assess your risk of exposure to zoonotic diseases based on your interactions with animals and environment. This tool helps you understand One Health principles in practice.
One Health Recommendations
More than 60% of all human infectious diseases come from animals. That’s not a guess-it’s data from the World Health Organization and the World Organisation for Animal Health. Ebola, HIV, COVID-19, avian flu, Lyme disease, rabies-all of them started in animals before jumping to people. And the next one? It’s already out there, hiding in the wild, in livestock, or in the insects buzzing around our cities. The old way of handling this-separate teams for human health, animal health, and environmental protection-is failing. We’re seeing outbreaks spread faster, harder, and with more chaos than ever. The solution isn’t more hospitals or better drugs. It’s One Health.
What One Health Really Means (And Why It’s Not Just a Buzzword)
One Health isn’t a fancy term for teamwork. It’s a fundamental shift in how we think about disease. It says: you can’t protect people without protecting animals and the environment they share. A sick cow in Kenya, a mosquito breeding in a drained swamp in Indonesia, a bat roosting in a newly cleared forest in Brazil-these aren’t isolated events. They’re warning signs. And they’re all connected. The idea isn’t new. Veterinarians and doctors have known for over a century that animal diseases can infect humans. But it wasn’t until the 2000s, after SARS and H1N1, that governments started treating it like a system problem. After COVID-19, the WHO, FAO, and WOAH launched a joint plan: 2021-2026 is the global push to make One Health real. Not just a policy paper. Real coordination. Real data sharing. Real funding.The Three Types of Zoonotic Threats You Can’t Ignore
Not all animal-to-human diseases are the same. Understanding the difference helps you see where to focus.- Endemic zoonoses are the quiet killers. Rabies, brucellosis, leptospirosis-they’re everywhere. They don’t make headlines, but they kill tens of thousands every year, mostly in poor rural communities. In Kenya, farmers lose livestock and children get sick from raw milk. In the U.S., people get sick from undercooked ground beef or contact with pet reptiles.
- Epidemic zoonoses explode suddenly. Think 2009 H1N1 flu. It started in pigs, jumped to people, and spread globally in weeks. These are the ones that shut down borders and trigger panic. They’re rare, but devastating.
- Emerging zoonoses are the ticking bombs. MERS, Nipah virus, new strains of avian flu-they’ve been around, but now they’re spreading faster because of deforestation, climate change, and global trade. A virus that once lived in a single bat colony in a remote forest is now in a live animal market in a city of 10 million people.
How Zoonotic Diseases Actually Spread (It’s Not Just ‘Contact’)
People think “zoonotic” means petting a dog or eating meat. It’s way more complex. Scientists break transmission into four types:- Orthozoonoses: Direct spread. Like rabies from a dog bite.
- Cyclozoonoses: Need multiple hosts. Tapeworms that live in dogs, then infect humans through contaminated soil.
- Metazoonoses: Require vectors. Lyme disease from ticks, West Nile from mosquitoes.
- Saprozoonoses: Spread through the environment. Hantavirus from rodent droppings in dusty barns, anthrax from spores in soil.
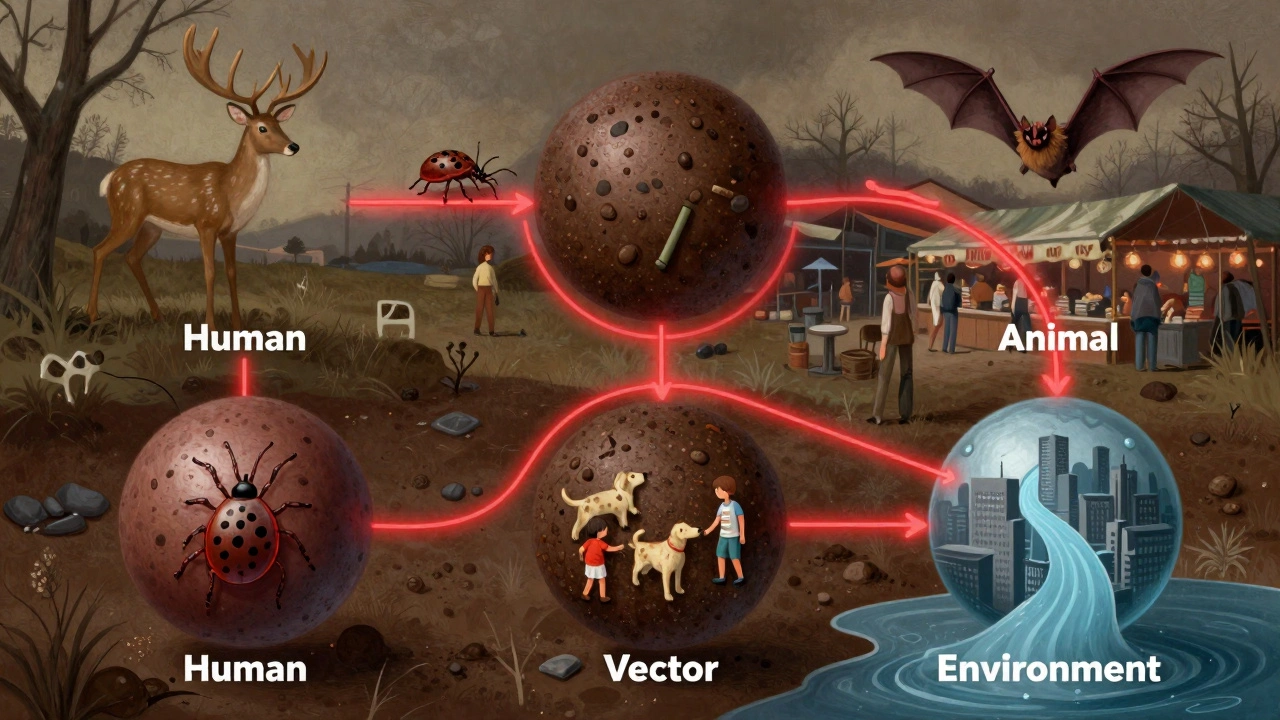
Why Siloed Systems Are Falling Apart
For decades, human health, animal health, and environmental agencies operated like separate islands. Each had its own budget, its own data systems, its own bosses. When a disease jumped from animals to people, no one knew who was responsible. Who tracks sick pigs? The agriculture department. Who tracks sick people? The health ministry. Who monitors forest loss? The environment agency. They didn’t share data. They didn’t even use the same file formats. The result? Delays. Confusion. Missed warnings. In Southeast Asia, a new avian flu strain showed up in poultry in 2021. The poultry farmers reported it. The vet agency took weeks to respond. The human health team didn’t get notified until five people were hospitalized. By then, the virus had spread to three provinces. A 2023 survey of 147 One Health practitioners found that 73% of countries still struggle with incompatible data systems. In low-income regions, 89% lack enough trained staff to monitor animal diseases in remote areas. And 52% of programs collapse after initial funding ends because there’s no permanent structure to keep them going.Where One Health Actually Works (And Why)
It’s not all doom and gloom. There are real wins. In Zanzibar, Tanzania, a simple program cut rabies deaths by 95%. How? They trained local animal health workers to vaccinate dogs door-to-door. At the same time, they taught schools about bite prevention and gave free post-exposure shots to people who got bitten. The vet team and the health team shared a single database. They held monthly meetings. They shared budgets. It worked because they treated it as one problem, not three. In Kenya, a county integrated livestock vaccination records with human health clinics. When a cow tested positive for brucellosis, the system automatically flagged people who drank that cow’s milk. Doctors started testing those patients early. Within two years, human cases dropped 37%. The common thread? Community involvement. In both cases, they didn’t send in experts from the capital. They trained local people-farmers, village health workers, schoolteachers-to be the eyes and ears. These are the people who notice a dead bat in the yard, a sick goat, or a sudden drop in chicken eggs. They’re the first line of defense.The Real Cost of Doing Nothing
The World Bank estimates zoonotic outbreaks cost the global economy $60-100 billion every year. That’s not just medical bills. It’s lost tourism, closed markets, supply chain chaos, and panicked travel bans. The 2009 H1N1 pandemic cost over $50 billion in the U.S. alone. COVID-19? Over $16 trillion globally. Dr. Mark Smolinski of Ending Pandemics says investing $10 per person per year in One Health could prevent pandemics that cost trillions. That’s $80 billion a year for the entire world. Compare that to the $16 trillion lost to one pandemic. The math is obvious. Yet less than 5% of global health funding goes to preventing zoonotic diseases at the animal source. Most money still goes to treating sick people in hospitals-after the outbreak has already happened. That’s like buying fire extinguishers after the house burns down.
What’s Changing Now (And What’s Coming)
In 2022, the U.S. launched its National One Health Framework, forcing 12 federal agencies-from the CDC to the USDA to the EPA-to coordinate. In Europe, every country now has a national One Health action plan. The WHO and FAO released a new risk-assessment tool in 2023 that helps countries predict where the next spillover might happen. The biggest shift? AI. The World Bank is funding a $50 million project to build AI models that scan satellite images, animal trade records, climate data, and human case reports to predict where a disease is likely to jump next. Think of it like a weather forecast-for pandemics. By 2030, the Global Health Security Index predicts 90% of rich countries and 60% of poorer ones will have permanent One Health units. That’s not because they’re being nice. It’s because they’re scared. And smart.How to Get Started (Even If You’re Not a Government)
You don’t need a national budget to start thinking like One Health. Here’s how:- Map your local interface. Where do people, animals, and nature overlap? A backyard chicken coop? A wet market? A park with deer? A farm near a river?
- Find the local players. Who talks to animals? The vet. Who talks to people? The clinic. Who manages land? The city parks department or a conservation group. Call them. Have coffee. Ask: “Have you seen anything unusual?”
- Start small. Track one disease. Rabies? Lyme? Salmonella from eggs? Collect simple data: number of sick animals, number of sick people, where they live, what they had in common.
- Share it. Use a free Google Sheet. Email it. Present it at a town meeting. Make it visible.
- Push for policy. Ask your local health department to include animal health data in their reports. Ask your city council to fund community animal health workers.





